Phase 1/2a Clinical Trial for Muscle Atrophy
Clinical Trial Listing: An open-label dose escalation study to assess the safety and tolerability of IMM01-STEM in participants with muscle atrophy related to knee osteoarthritis
Target: Muscle atrophy; sarcopenia
Treatment: IMM01-STEM
Status: Active
ClinicalTrials.gov Identifier: NCT05211986
Every individual experiences a decline in immune system health as they age, which contributes to degenerative conditions throughout the body, including muscle atrophy. Age-related muscle atrophy (sarcopenia) and muscle wasting from disuse or disease significantly decreases quality of life. Mitigating the loss of muscle and improving muscle recovery remain unmet medical needs. Our goal is to utilize a multi-active biologic (IMM01-STEM) as a means of driving specific immune responses to overcome manifestations of age-related disease.
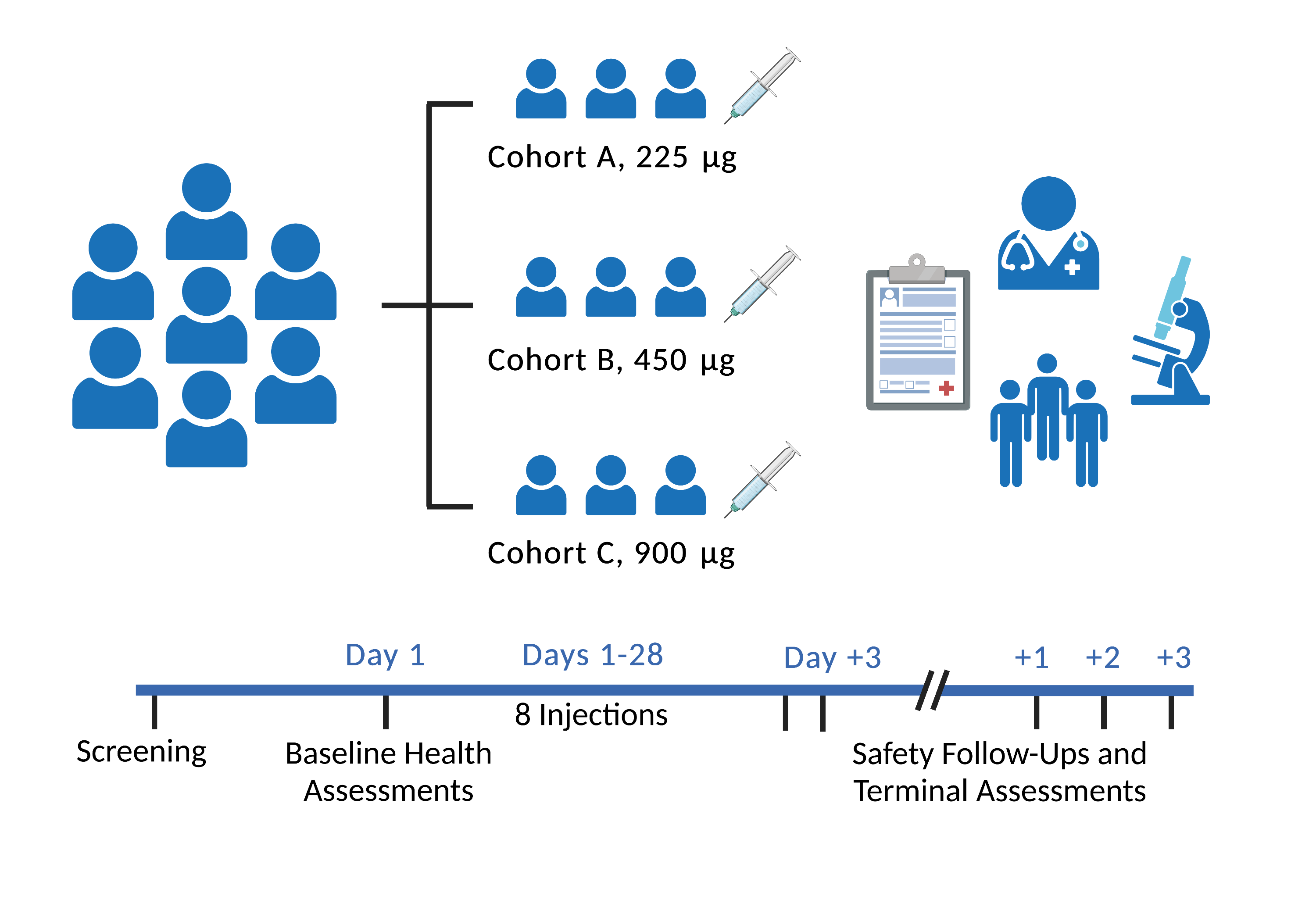
Immunis is completing a Phase 1/2a clinical trial examining the safety, tolerability and preliminary efficacy of IMM01-STEM treatment in sarcopenic elderly with knee osteoarthritis. The FDA-approved Phase 1/2a clinical trial is a single-center, open-label, dose-escalation study to assess safety, tolerability and preliminary efficacy of IMM01-STEM in elderly participants with muscle atrophy related to knee osteoarthritis. IMM01-STEM was administered via intramuscular (im) injection twice per week for 4 weeks.
There have been no reports of serious adverse events (SAEs) associated with our IMM01-STEM treatment in humans. Initial analyses show striking improvements in quality-of-life measurements related to pain, stiff, and physical function. In the 6-min walk test, the patients increased their walking distance and achieved a clinically meaningful improvement in gait speed, an encouraging suggested functional efficacy signal. The team is preparing a manuscript detailing the full results of the investigation.
The Phase 1/2a was conducted at the University of California Irvine Medical Center, UCI Health. UCI Health is part of the California Institute of Regenerative Medicine's Alpha Clinic Network.