What causes sarcopenia?
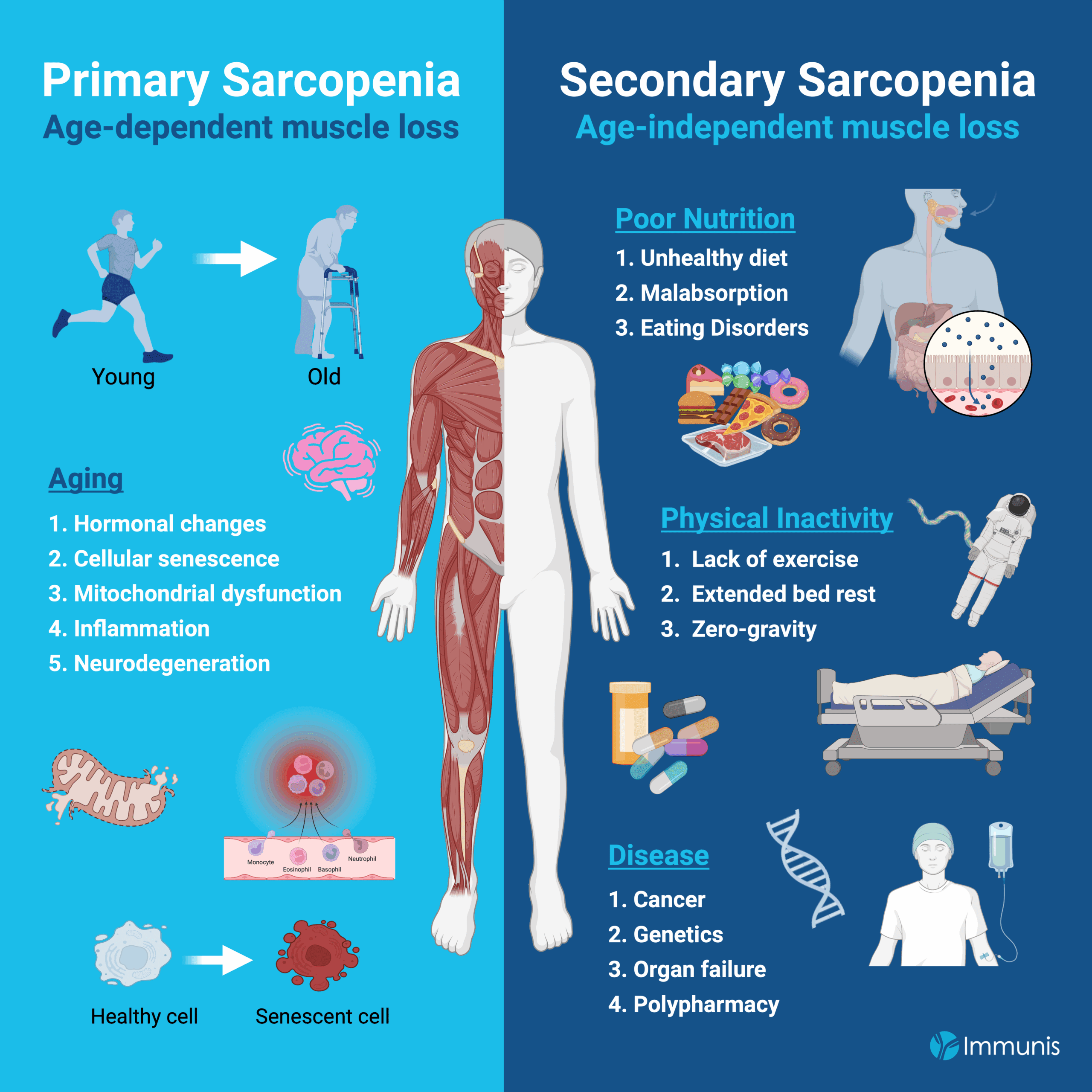
There are two forms of sarcopenia, primary sarcopenia and secondary sarcopenia. Aging is the greatest risk factor for developing primary sarcopenia,1 but a variety of age-independent factors such as poor nutrition, physical inactivity, and disease can also negatively affect muscle structure and function, known as secondary sarcopenia.2
Drivers of Primary Sarcopenia
100% of people develop primary sarcopenia as they get older. This type of muscle loss is driven by aging and a variety of age-dependent factors including changes in hormone levels, cellular senescence, mitochondrial dysfunction, inflammation, and neurodegeneration.3
-
Hormonal changes
Over time, hormonal changes can alter muscle size and function. For example, testosterone is important for protein synthesis and muscle maintenance, and there is a strong association between muscle atrophy and low testosterone levels.2 Between the ages of 20-30, testosterone levels gradually decline. Around the age of 50, women enter menopause, marked by a significant decrease in estrogen, growth hormone, and testosterone. A lesser known, but similar process also occurs in men called andropause, which is characterized by reduced testosterone levels. Thus, hormones like testosterone that fluctuate with age and are important for muscle regeneration can be a driving factor that affects muscle performance.
Another example is the stress hormone, cortisol. Cortisol promotes muscle breakdown and is shown to exacerbate muscle loss and reduce strength during bed rest. One study showed that healthy young men who were administered hydrocortisone during a month-long bedrest had a greater loss in lean mass in their legs compared to those who were also on bedrest, but did not receive hydrocortisone.511β-HSD1, responsible for converting cortisone to active cortisol, has been shown to regulate protein metabolism and influence the size of muscle fibers. Additionally, there is a negative correlation between individuals with a high expression of the enzyme 11β-HSD1, and grip strength, further supporting the role of the cortisol hormone in muscle function.5
-
Cellular senescence
Aging results in a time-dependent accumulation of molecular and cellular damage to the body, including the immune cells. Senescence is when these cells can no longer respond appropriately to the complex regulatory signals of their environment; thereby causing an imbalance of inflammatory and anti-inflammatory responses that can exacerbate muscle damage.3,6
Satellite cells are skeletal muscle stem cells essential for muscle repair, but their number and regenerative capacity decline with age.7 When there is muscle damage, either by injury or exercise, satellite cells activate, proliferate, and fuse with muscle fibers to repair the tissue. However, senescent satellite cells have reduced proliferative capacity, impairing muscle regeneration and contributing to sarcopenia.8
-
Inflammation and inflammaging
Chronic, low-level inflammation is common with age (also known as inflammaging) and negatively impacts skeletal muscle. Inflammaging results in the production and release of toxic cellular secretions that causes a breakdown in muscle fibers and slows protein synthesis necessary for muscle regeneration.3 The persistence of damage signals recruits additional immune cells in a positive inflammatory feedback loop that exacerbates sarcopenia.9 Thus, inflammation is both a cause and a consequence of the progression of sarcopenia.1
-
Mitochondrial dysfunction
Mitochondria are specialized energy-producing structures within our cells. As we age, there is an accumulation of changes in the mitochondria, which impairs energy production and promotes stress that damages our muscle.7 Damaged mitochondria can trigger the self-destruction of cells, speeding up the breakdown of muscle fibers.10,11 Studies of muscle tissue from over 100 older adults have found that those with sarcopenia have mitochondria with disrupted metabolism and repair mechanisms, which exacerbates muscle atrophy and weakness.12,13 Therefore, defects in mitochondria have significant consequences on muscle health.
-
Neurodegeneration
After the age of 60, humans lose about 25-50% of their motor neurons, known as neurodegeneration.14,15 This age-related reduction in neurons results in fewer connections between the muscle and the nervous system that can trigger muscle movement. Without sufficient stimulus of the muscle tissue by the neurons, older individuals have a more challenging time with managing their motor skills.
Drivers of Secondary Sarcopenia
Secondary sarcopenia is muscle loss that is independent of age and in some cases, it can exacerbate primary sarcopenia. Major contributors to secondary sarcopenia are poor nutrition, physical inactivity and disease.
A. Poor Nutrition
An unhealthy diet, malabsorption, or eating disorders can prevent the body from obtaining the necessary proteins and nutrients to maintain and promote muscle growth.16,17
-
Unhealthy Diet
A fatty diet can promote obesity, which creates a higher risk of disability and disease. Sarcopenia and obesity are closely linked, a condition called sarcopenic obesity, where the prevalence of one condition worsens the other. Obesity results in muscle loss by promoting a pro-inflammatory tissue environment that is less conducive for building muscle and in some case, encourages skeletal muscle breakdown.18 Muscle is a highly metabolic tissue that constantly burns calories, even at rest. Therefore, sarcopenic obesity stemming from an unhealthy diet creates a vicious cycle where muscle loss consequently slows metabolism, thereby further contributing to a gain in fat mass.19
Please refer to our resource titled “Sarcopenia and Obesity” for additional information.
-
Malabsorption
People with gastrointestinal issues may have malabsorption of the nutrients in the food they eat. This means that even if a person is consuming a recommended diet, their body is unable to properly metabolize food to provide key materials used by their cells. The insufficient absorption of beneficial amino acids is detrimental because they are necessary for muscle regeneration, and when limited, can promote sarcopenia.16,20
-
Eating Disorders/Malnutrition
More than 40% of people aged 70+ consume less than the recommended daily amount of protein (1-1.5g/kg bodyweight) and are expected to have an increased prevalence of sarcopenia.21 Insufficient consumption of key nutrients like protein, vitamin D, and calcium compromises the preservation of muscle mass, strength, and function. One of the challenges for older individuals is that they may have dietary restrictions that prevent them from being able to consume enough protein to sustain muscle mass, much less promote muscle growth. Decreased appetite is observed in 64% of patients during hospitalization and in 28% following discharge.22 Eating disorders in which the body is deprived of food, such as anorexia nervosa and bulimia, decreases muscle mass and reduces physical performance by up to 50% compared to healthy controls.23 Due to the relatively slow turnover rate of skeletal muscle, roughly ~1-2% per day, insufficient protein consumption likely accelerates muscle loss caused by disuse atrophy.3 Additionally, muscle fibers contain vitamin D receptors, and there is evidence suggesting that vitamin D is important for skeletal muscle regeneration after injury because of its role in the integrity of satellite cells (muscle stem cell).17
B. Physical Inactivity
Muscle is a true “use it or lose it” type of tissue. A study by the Centers for Disease Control and Prevention reported that physical inactivity and poor diet was the second leading cause of preventable deaths from 1980–2002.24 Additionally, the US Department of Health and Human Services reports that people are at a greater risk of all-cause mortality from a lifetime of inactivity.25 Thus, a sedentary lifestyle not only accelerates the decline in skeletal muscle, it can also significantly shorten life expectancy.26
A lack of exercise, extended bedrest, and micro-gravity are a few of the scenarios in which there is limited exertion of muscle, which can promote muscle atrophy (aka muscle wasting).
-
Lack of exercise
Simply put, working out helps to build and maintain muscle and the converse is true, where inactivity diminishes muscle size and strength. Please refer to our sarcopenia resource titled “Is there a treatment or a cure?” for additional information on how to build muscle with resistance exercise.
-
Extended bedrest
Long-term bedrest can be detrimental to health because muscles are rarely being used. For example, healthy volunteers who participated in periods of bed rest had reduced muscle quantity, strength, and aerobic performance.3 Other instances of extended bedrest include hospitalizations. For example, people who receive a hip replacement surgery show a reduction in muscle area, which can persist for up to 3.5 years post-surgery.3
-
Microgravity
Additionally, astronauts are at a significant risk of losing muscle and bone mass during space expeditions because the micro/zero-gravity conditions limit the need for muscle exertion. For this reason, NASA has been optimizing their strength training regimes for astronauts to minimize muscle loss during their space missions.27
Ultimately, not using your muscles will cause them to shrink and this can significantly shorten life expectancy and quality of life.26
C. Disease
-
Genetics
Genetic factors can predispose individuals to sarcopenia by influencing key determinants of muscle mass, function, and regenerative capacity. For example, genetic deletions can accumulate in muscle fibers over time, significantly limiting energy production to support muscle growth and cause the destruction of muscle cells.28 However, the impact of these genetic defects may not become apparent until the individual is older, when cells have accumulated many mutations.
-
Cancer, organ failure, and polypharmacy
Other diseases like cancer and advanced organ failure can also negatively impact muscle due to the increased likelihood of long-term hospitalization. In many cases, the ICU or long-term care facilities treat their patients with multiple pharmaceutical drugs simultaneously (polypharmacy) to combat their disease.4 However, toxicity from the repeated use of drug cocktails can further weaken the body. There is a strong correlation between polypharmacy and sarcopenia, which makes the case for slowly deprescribing medications to mitigate their deleterious effects on muscle.4 Researchers assessed the effect of deprescribing drugs in the hospital setting for sarcopenic individuals aged 65+ recovering from stroke and taking at least five prescription medications. In those who underwent deprescribing, there was an improvement in independent measure-motor scores upon discharge and their likelihood of discharge also increased.4 While pharmaceuticals can be life-saving drugs, studies suggest that too many medications can cause more harm than good.
In summary, sarcopenia presents in two forms: primary sarcopenia, which is driven by aging, and secondary sarcopenia, which arises from age-independent factors such as inadequate nutrition, physical inactivity, and underlying disease. While aging remains the predominant risk factor for primary sarcopenia, it is essential to recognize that potentially modifiable lifestyle and health factors can independently compromise muscle mass and function, leading to secondary sarcopenia.
Additional Resources
-
Grima-Terrén, M. et al. Muscle aging and sarcopenia: The pathology, etiology, and most promising therapeutic targets. Molecular Aspects of Medicine 100, 101319 (2024).
-
Cruz-Jentoft, A. J. et al. Sarcopenia: revised European consensus on definition and diagnosis. Age and Ageing 48, 16–31 (2019).
-
Damanti, S. et al. Acute Sarcopenia: Mechanisms and Management. Nutrients 16, 3428 (2024).
-
Chung, J. Y., Kim, S.-G., Kim, S. H. & Park, C. H. Sarcopenia: how to determine and manage. Knee Surgery & Related Research 37, 12 (2025).
-
Hassan-Smith, Z. K. et al. Gender-Specific Differences in Skeletal Muscle 11β-HSD1 Expression Across Healthy Aging. J Clin Endocrinol Metab 100, 2673–2681 (2015).
-
Isobe, K., Nishio, N. & Hasegawa, T. Immunological aspects of age-related diseases. World J Biol Chem 8, 129–137 (2017).
-
Nunes-Pinto, M. et al. Sarcopenia and the biological determinants of aging: A narrative review from a geroscience perspective. Ageing Research Reviews 103, 102587 (2025).
-
He, Y. et al. Cellular Senescence in Sarcopenia: Possible Mechanisms and Therapeutic Potential. Frontiers in Cell and Developmental Biology 9, (2022).
-
Liang, Z. et al. Inflammaging: The ground for sarcopenia? Exp Gerontol 168, 111931 (2022).
-
Ferri, E. et al. Role of Age-Related Mitochondrial Dysfunction in Sarcopenia. Int J Mol Sci 21, 5236 (2020).
-
Bellanti, F., Lo Buglio, A. & Vendemiale, G. Mitochondrial Impairment in Sarcopenia. Biology (Basel)10, 31 (2021).
-
Migliavacca, E. et al. Mitochondrial oxidative capacity and NAD+ biosynthesis are reduced in human sarcopenia across ethnicities. Nat Commun 10, 5808 (2019).
-
Dao, T. et al. Sarcopenia and Muscle Aging: A Brief Overview. Endocrinol Metab (Seoul) 35, 716–732 (2020).
-
Rygiel, K. A., Picard, M. & Turnbull, D. M. The ageing neuromuscular system and sarcopenia: a mitochondrial perspective. The Journal of Physiology 594, 4499–4512 (2016).
-
Tomlinson, B. E. & Irving, D. The numbers of limb motor neurons in the human lumbosacral cord throughout life. Journal of the Neurological Sciences 34, 213–219 (1977).
-
Liu, S., Zhang, L. & Li, S. Advances in nutritional supplementation for sarcopenia management. Front. Nutr. 10, (2023).
-
Adegoke, O. A. J., Huang, Y., Fu, X. & Mora, S. Editorial: Nutrition in the Regulation of Muscle Development and Repair. Front Physiol 13, 853007 (2022).
-
Li, C. et al. Pathogenesis of sarcopenia and the relationship with fat mass: descriptive review. J Cachexia Sarcopenia Muscle 13, 781–794 (2022).
-
Argilés, J. M., Campos, N., Lopez-Pedrosa, J. M., Rueda, R. & Rodriguez-Mañas, L. Skeletal Muscle Regulates Metabolism via Interorgan Crosstalk: Roles in Health and Disease. J Am Med Dir Assoc 17, 789–796 (2016).
-
Mielgo-Ayuso, J. & Fernández-Lázaro, D. Nutrition and Muscle Recovery. Nutrients 13, 294 (2021).
-
He, W., Connolly, E. D., Cross, H. R. & Wu, G. Dietary protein and amino acid intakes for mitigating sarcopenia in humans. Critical Reviews in Food Science and Nutrition 65, 2538–2561 (2025).
-
Reijnierse, E. M. et al. The Association between Parameters of Malnutrition and Diagnostic Measures of Sarcopenia in Geriatric Outpatients. PLoS One 10, e0135933 (2015).
-
Casper, R. C. Might Starvation-Induced Adaptations in Muscle Mass, Muscle Morphology and Muscle Function Contribute to the Increased Urge for Movement and to Spontaneous Physical Activity in Anorexia Nervosa? Nutrients 12, 2060 (2020).
-
Mokdad, A. H. Actual Causes of Death in the United States, 2000. JAMA 291, 1238 (2004).
-
U.S. Department of Health and Human Services. Physical Activity Guidelines Advisory Committee Report, Part A: Executive Summary. Nutrition Reviews 67, 114–120 (2009).
-
Booth, F. W., Laye, M. J. & Roberts, M. D. Lifetime sedentary living accelerates some aspects of secondary aging. Journal of Applied Physiology 111, 1497–1504 (2011).
-
Astronaut Exercise - NASA. https://www.nasa.gov/missions/station/iss-research/astronaut-exercise/ (2024).
-
Hiona, A. et al. Mitochondrial DNA Mutations Induce Mitochondrial Dysfunction, Apoptosis and Sarcopenia in Skeletal Muscle of Mitochondrial DNA Mutator Mice. PLOS ONE 5, e11468 (2010).