In the past few decades, the number of overweight and obese individuals in the U.S. has skyrocketed, especially in older adults. According to the National Center for Health Statistics, the percentage of obese Americans aged 65+ has doubled from 20% to 40% for both men and women.2 Obesity coupled with sarcopenia, known as sarcopenic obesity, is characterized by an increase in fat and reduction in muscle mass and function.3–5 With sarcopenic obesity, the skeletal muscle can exhibit a variety of physiological changes including muscle shrinkage, altered muscle fiber composition, increased intramuscular fat, and reduced muscle quality, which is the ability for muscle to produce a force relative to its size.2 Roughly 40-80 million people worldwide are currently affected by SO, and this number is projected to reach up to 200 million within the next four decades.6
The individual health risks of sarcopenia and obesity have been well-documented, but combined, they worsen clinical health outcomes.2,7 For example, sarcopenic obesity (SO) (BMI 30 kg/m2) poses a greater risk for frailty, type 2 diabetes, metabolic diseases, cardiovascular disease, and all-cause mortality.1,2,8 While individuals who are overweight (BMI ≥25 kg/m²) and exhibit sarcopenia (SOW) do not necessarily meet the criteria for sarcopenic obesity, they are at elevated risk of progressing to this condition.
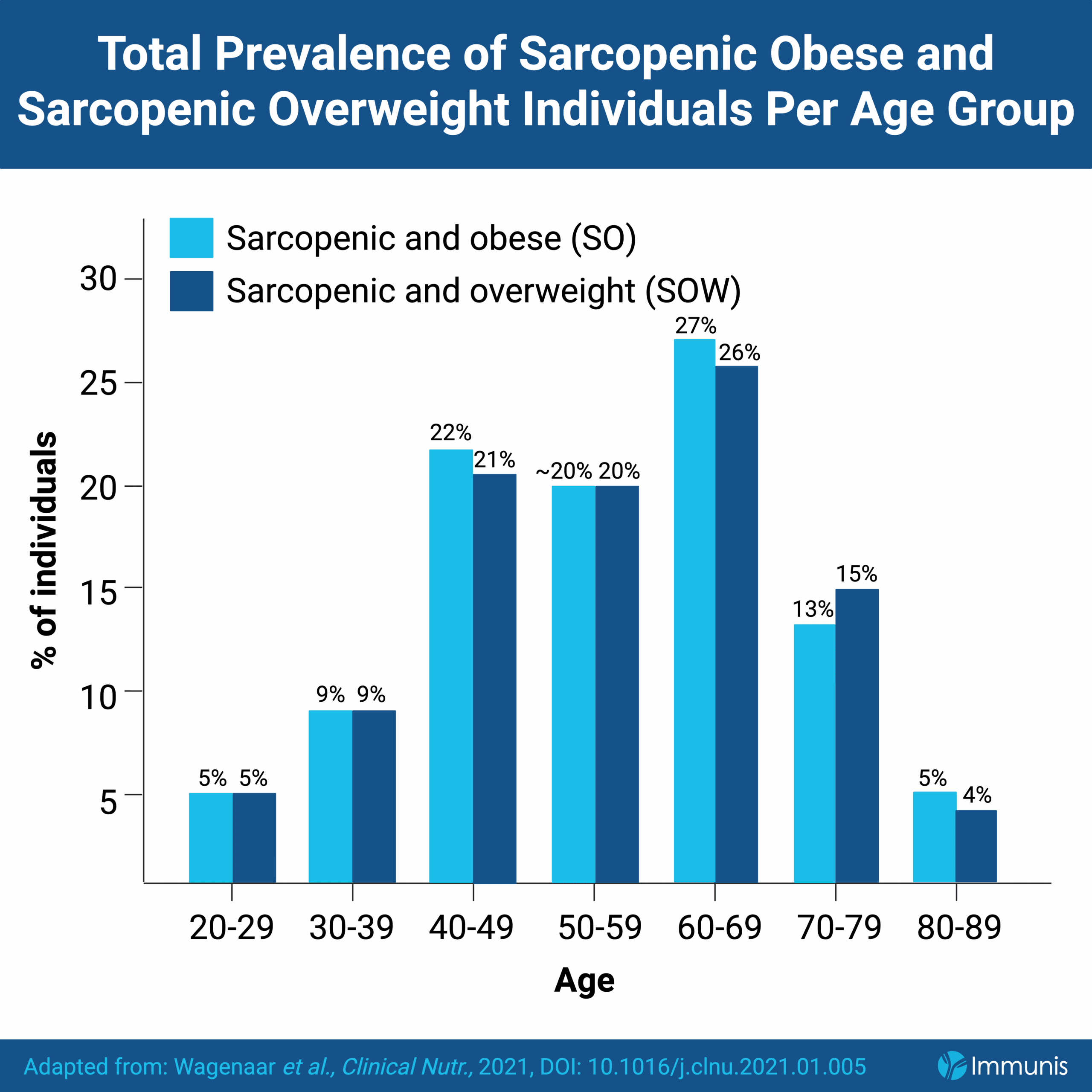
The Lifelines population-based cohort study, which evaluated data from over 100,000 men and women aged 18-90, showed a positive correlation between the prevalence of SO and SOW with age.1 Of the total number of subjects with SO and SOW, 69% and 67%, respectively, were between the ages of 40-69, while the prevalence was only 14% for those aged 20-39.1 As both SO and SOW are observed more frequently in middle-aged adults, there is a need to develop treatments for SO that cater to these age groups and more importantly, there is a need to design interventions that prevent the development of SOW into SO. Unfortunately, SO and SOW are often underdiagnosed due to heterogeneity in diagnostic criteria, adding to the challenge of developing effective prevention plans.2,7
A balanced, high-protein diet and consistent resistance exercise are the recommended approaches to losing weight and gaining muscle, but the variability in SO produces inconsistent outcomes. Differences in age, the presence of chronic conditions, current physical capabilities, and other factors require individualized therapeutic strategies, which may be challenging to formulate.9 Treatment plans for SO should not only draw on expertise from clinical nutrition, endocrinology, metabolism, geriatric medicine, and sports medicine, but also be sustainable for the patient long-term.9
Another treatment option is bariatric surgery, which has been successful in promoting weight loss and improving comorbidities in patients aged 60 and up. However, it is an invasive procedure that also increases the risk of surgical complications, morbidity, and mortality, while doing nothing to help promote muscle regeneration.10
More recently, pharmacotherapies like GLP-1 receptor agonists have become popularized for their ability to achieve rapid weight loss results. $71.7 billion was spent on GLP-1 drugs in 2023 alone, over a 500% increase from 2018.11 These therapies may appear on the surface to be an ideal approach to tackling conditions like SO. However, 25-40% of the total weight lost from these drugs can be attributed to a decrease in lean muscle mass, not fat.12,13 Thus, individuals taking GLP-1s over an extended period may experience a decline in muscle size and function over time. As there are no FDA-approved pharmaceutical interventions to promote muscle regeneration, GLP-1s may cause issues for elderly individuals who are already at a greater risk of muscle loss. Additionally, little is known about the long-term health effects of GLP-1s. While GLP-1s may address one aspect of sarcopenic obesity (fat loss), they also exacerbate another (muscle loss). For this reason, integrating multimodal interventions or developing novel drugs that tackle both concerns of sarcopenic obesity are necessary to maximize long-term health outcomes.
Immunis is seeking to address these clinical challenges with the investigational secretome, IMM01-STEM, which may reduce fat, while preserving muscle. In preclinical data published in Aging Cell, IMM01-STEM treatment in aged mice increased whole-body lean mass and reduced fat mass, while increasing muscle size, enhancing muscle regeneration, and improving muscle function.14 Immunis’ Phase 1/2a clinical trial showed no adverse events attributable to IMM01-STEM, with a suggestion of enhanced muscle function. Immunis is also conducting a Phase 2 clinical trial to test the efficacy of IMM01-STEM in combating sarcopenic obesity, in the hopes of increasing mobility and independence.